Plating Q&A and Vocabulary
Plating film |
Plating solution |
Materials and Process |
Vocabulary |
Please click on the item.
About plating film |
What is the difference between electroplating and electroless plating?
In both plating methods, nickel ions (Ni2+) in the plating tank are reduced by receiving electrons and deposited as metal on the surface of the material.
Electroplating is done by generating a potential difference between electrodes using an external power source and providing electrons from the cathode to deposit metal, while electroless plating is done by using a chemical reaction (a reaction in which a substance is oxidized) to provide electrons to metal ions to deposit metal. (Electroless plating is also called chemical plating.)
Electroplating is efficient because it receives electrons directly from the cathode, but
- It is necessary to firmly establish contact between the jig and the item to improve the flow of electricity.
- If there is a shield between the cathode and anode, it will be electrically shaded and the deposition properties of that part will be poor.
- The current density at the edge part will be high due to the concentration of the electric field, which can cause problems such as the plating film thickness being different from other areas.
On the other hand, electroless plating uses a chemical reaction, so the part in contact with the plating solution will react uniformly, making it possible to obtain a uniform film thickness. The structure of the jig can also be simpler than that of electroplating.
However, even within the same plating tank, if there is a localized uneven temperature distribution, poor circulation of the solution, or if new plating solution is not constantly supplied, the deposition in that area will deteriorate, so care must be taken. The key to obtaining a good film is how to maintain a uniform concentration and temperature throughout the entire plating tank.
Another feature of electroless nickel plating is that a reducing agent is used as a raw material for the plating solution other than nickel, and some of the elemental components are incorporated into the film, forming an alloy film such as Ni-P or Ni-B. Sodium hypophosphite, DMAB (dimethylamine borane), hydrazine, etc. are used as reducing agents, and films with different properties can be obtained from each of them.
Why does heat treatment harden plating films?
This is because the film, which was amorphous at the time of deposition, changes to crystalline.
Low phosphorus films (SE-797) and KANIBORON (Ni-P-B), which are crystalline at the time of deposition, have a higher hardness at the time of deposition than medium and high phosphorus films.
For example, with common medium and high phosphorus plating solutions such as SE-660, the hardness begins to increase from the late 200°C, and is at its highest between the late 300°C and 400°C (around Hv900). However, when baking in an air atmosphere, discoloration occurs due to oxidation of the film surface, so care must be taken with exterior parts.
For materials such as aluminum and aluminum alloys that may become embrittled at high temperatures, it is better to select low phosphorus films (SE-797) or KANIBORON (Ni-P-B), which can obtain hardness without heat treatment.
What are the advantages of Kanigen plating (Electroless Nickel plating) compared to electro Chromium plating?
1. A uniform film is obtained.
2. Relatively few pinholes.
3. Good adhesion to the product.
These are some of the features.
To bring out the best in both films, chromium plating is sometimes applied on top of electroless plating.
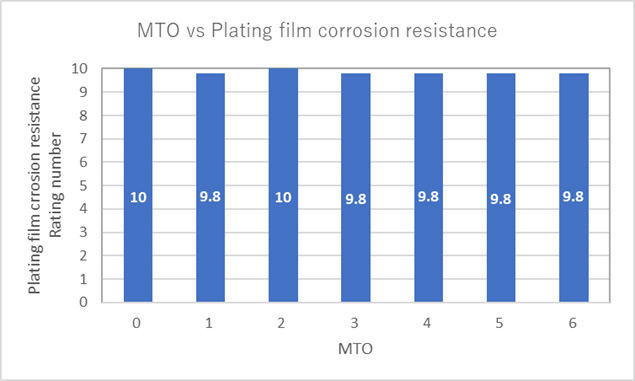
How long does Kanigen plating last in a salt spray test?
Basically, it depends on the type of plating film and the plating thickness.
If it's for a few hours, we think it's possible to maintain a rating number of 9.8 or higher even with a plating film thickness of 3 to 5 μm.
However, the results will vary depending on the plating conditions (pretreatment method, product shape, product surface condition, etc.), so we recommend that you evaluate it in advance. (*When evaluating with a test piece, care must be taken as the conditions may differ from those of the product.)
For this reason, we recommend that you perform the plating on something made of the same material and with the same surface condition as the product and then perform the evaluation. If you have specific evaluation conditions, please contact us.
Reference example: SE-660
[Evaluation conditions]
Salt spray test based on JIS standard H8502-1999
|
Spray solution |
: 50g/L sodium chloride |
|
Spray time |
: 24hours |
|
Spray chamber temperature |
: 35±2℃ |
|
Test piece |
: 1dm2 test piece (SPCC material) |
|
Coating thickness |
: Approximately 5μm |
※No heat treatment or post-treatment
[Evaluation results] Rating number = 10 is considered to be no rust.
What are the characteristics of KANIBORON (Ni-P-B)?
It has excellent sliding properties in a lubricated environment. In addition, high hardness (Hv750 or higher) can be achieved through low-temperature heat treatment (200°C).
Please see the product introduction for details.
What are the features of CERAMIC KANIGEN (Ni-P+SiC)?
It is a composite film of SiC, and has abrasion resistance (Hv1000 or more) that is higher than that of hard chromium. It is most suitable for sliding parts that are subject to severe wear such as being scraped by a file.
Please see the product introduction for details.
Please tell me the principle of CERAMIC KANIGEN (Ni-P+SiC)
SiC is co-deposited into the Ni-P film by free fall due to gravity. Almost no co-deposition occurs on the sides and bottom of the product. In order to deposit SiC evenly over the entire surface, some ingenuity, such as the use of a rotating jig, is required.
What are the features of KANIFLON(Ni-P+PTFE)?
It is possible to reduce the coefficient of friction and obtain lubricity even in a non-lubricated environment.
Please see the product introduction for details.
Please explain the principle of KANIFLON(Ni-P+PTFE)
The principle is that PTFE is electrostatically adsorbed onto the surface of the part using a surfactant. PTFE is uniformly dispersed and co-deposited on the top, bottom, and sides of the part.
What are the features of KANIBLACK(black film)?
When an item is immersed in a plating bath, a black film is instantly formed on the surface, and over time the black film grows continuously, resulting in an autocatalytic plating that can achieve the required thickness. It has excellent low reflectivity and light absorption properties, and is used for optical components and printing equipment parts. Please see the product introduction for details.
Is it possible to use KANIBLACK on exterior surfaces?
KANIBLACK absorbs light by depositing a film in a broccoli-like shape. This broccoli-like unevenness is weak, so it is basically impossible to use it on exteriors. However, since the unevenness of "KANIBLACK II" is strong, it can be used in places where no force is applied.
What are the characteristics of KANIHASUTE?
This is an electroless plating film of an alloy that combines high hardness at the time of deposition (Hv770) and high hardness in a high-temperature environment (Hv560 at 300℃). This is developed film by our company that enhances the sliding properties in a high-temperature environment while also providing sliding properties in a dry environment.
About plating solution |
Is the phosphorus content in electroless nickel plating solutions constant?
The phosphorus content in the film is not constant, as it is affected by the plating solution composition, plating tank temperature, pH, and number of turns (accumulation of phosphite ions and sulfate ions). In general, the following relationship holds:
- Plating temperature, pH: The higher these are the lower the phosphorus content.
- Number of turns: The older the plating solution, the higher the phosphorus content.
- Sulfur-based additives: the addition of sulfur additives reduces the phosphorus content.
For continuous plating solutions, after the plating solution is prepared, the phosphorus content will gradually increase when the plating solution is used.
Therefore, the phosphorus content must be controlled within a certain range, for example, 10-12%.
What is the difference between pre-treatment pickling and Acid activation?
The process is the same.
Pickling: Pretreatment such as removing oxide films
Acid activation: Activating the surface of the item before plating
What is the relationship between plating speed and pH/temperature?
Generally, the following relationship holds:
1. pH: the higher the pH, the faster the plating speed.
2. Temperature: the higher the temperature, the faster the plating speed.
However, raising either of these too much can cause the plating solution broken or become cloudy, so please use the solution within the ranges specified in the instruction manual. Please consult us if you wish to use the solution outside the ranges specified in the instruction manual.
Please tell me a simple way to calculate plating thickness.
The film thickness can be measured from the film weight.
To measure the film weight, please prepare an electronic balance that can measure to three decimal places (0.000).
*When calculating the surface area in "cm2"
Film thickness (μm) = film weight (g) / surface area (cm2) / specific gravity (g/cm3) x 10000
*When calculating the surface area in "dm2"
Film thickness (μm) = film weight (g) / surface area (dm2) / specific gravity (g/cm3) x 100
Film weight (g) = weight after plating (g) - weight before plating (g)
We have test pieces (0.189dm2) for measuring plating speed, so please contact us if you need one.
What is the preferred material for the plating tank?
SUS316 is the best, but SUS304 can also be used. SUS plating tanks can be passivated with nitric acid to form a passive film, making them ideal for electroless plating. Resin (PVC, etc.) plating tanks can also be used, but as they age, the surface becomes scratched and plating films become more likely to deposit in the plating tank.
When using electroless plating solution, are a filter pump and automatic analysis and replenishing device necessary?
The filter pump is necessary to reduce contaminants in the plating solution. The automatic analysis and replenishment device is also necessary to keep the pH and Ni concentration of the plating solution constant. (Of course, manual analysis is also possible.) Both pieces of equipment reflect our many years of know-how, and allow you to use the electroless plating solution more effectively and efficiently.
How should plating waste liquid be treated?
Basically, plating waste liquid are disposed by a specialist.
About materials and processes |
Is plating possible for any material?
Kanigen plating can be applied to all kinds of materials, including iron, special steel, stainless steel, aluminum, copper, The pre-treatment process for plating varies depending on the material. Please contact us if you have any special materials.
Is it possible to plate castings or die-cast products?
Yes, that is possible. However, the treatment method differs depending on the surface condition of the casting cavity, etc., so we will consult with you separately.
Is it possible to plate plastic?
Yes, it is. However, a different pre-treatment process is required than for metals. The basic process is "degreasing → etching → sensitization (Sn adsorption) → activation (Pd adsorption) → plating". Since it has a large thermal expansion coefficient, plating at a relatively low temperature is required. (SE-680, Ni-B, etc.)
Vocabulary |
Kanigen
Kanigen is a general term for electroless nickel plating, and our company name, Kanigen, is an acronym of C(K)atalytic, Nickel, Generation. Kanigen plating is a method of coating nickel-phosphorus alloys through a chemical reaction without using electricity.
Compared to other plating methods, Kanigen plating has the following characteristics:
- Uniform plating thickness
- Capable of plating non-conductors
- Capable of plating a wide range of metals
- Excellent adhesion, hardness, abrasion resistance, and corrosion resistance
Due to these characteristics, our technology is used in a wide range of fields, from familiar items such as automobiles, semiconductor parts, digital cameras, and computers to F1 cars, jet planes, and artificial satellites, as a functional plating that enhances the functions of industrial parts. This is also evidenced by the fact that Kanigen, which is also our company name, is referred to as "Kanigen plating," a general term for electroless plating.
SUMER
This is the general name for the plating solution developed by Japan Kanigen Co., Ltd. to allow electroless nickel plating to be performed easily anywhere.
It was named "Sumer" from the initials of Surface metalizer and began selling it.
Make-up
This is the process of preparing the plating solution in a specified ratio, adjusting the concentration and pH of each component, and preparing it in a state that is ready for plating. Of course, the term "bath make-up" is used not only for plating solutions, but also when preparing pretreatment solutions.
There are various methods for bath make-up, such as Blue Sumer, which uses one solution, and SE-660, which uses two or more solutions. (The standard bath make-up method is described in the instruction manual.)
In the case of Sumer solutions, the concentration and pH of each component are all prepared and analyzed at our manufacturing plant before shipping, so there is no need for detailed adjustments or analysis on the customer's line when making the bath. (There are some exceptions.)
Metal Turn Over (MTO)
MTO is an abbreviation of Metal Turn Over(It is sometimes called MTO)
In continuous plating solutions, it is used as a numerical value to know the aging of the solution. When the total additions of replacement metal that has added are equal to the total amount of metal originally in the bath, that’s one metal turnover.
For example, the nickel (sulfate) concentration of SE-660 24.75g/L during bath make up.
When an item is immersed in this plating solution and the plating reaction begins, the nickel concentration will gradually decrease. However, in reality, the concentration of nickel and reducing agent, and the pH decrease with the reaction, plating speed gradually decreases, and finally plating reaction stops, so it is impossible to plate until all 24.75 of nickel is used. Therefore, it is necessary to replenish NaOH or ammonia in the replenishment solution to compensate for the decrease in nickel, reducing agent, and pH, but if you replenish it halfway, you cannot judge how much has been used from the current nickel concentration. Therefore, the concept of number of turns is used.
Since we are focusing on nickel here, the number of turns is calculated from the amount of nickel salt replenishment solution replenished.
In the case of SE-660-A, the concentration of nickel sulfate is 450g/L, so 24.75÷450=0.055, that is, one turn is when 55ml is replenished for 1L of bath.
Even if the plating solution can be used for the same number of turns, if the nickel concentration at the time of make-up the bath is different, the surface area that can be treated will be different. Please note that if the plating solution is changed, the number of turns does not equal the capacity.
(dm2・μm)
It is a unit that expresses the amount of plating deposited by volume. and is used when calculating the unit price of plating and the required amount of replenishment solution.
When an area of 1 dm2 (100 cm2) is plated with a thickness of 1 μm, it is 1 μdm2 (μm x dm2).
If the plating solution is a medium-high phosphorus type, 1 gram of nickel sulfate can plate 2.88 μdm2.
We use 2.88 μdm2 as the experimental value, but please note that some manufacturers use theoretical values of 3.14 μdm2 or 3.0 μdm2. If the phosphorus content is about the same, the area that can be plated with 1 gram of nickel sulfate will be the same.
Also, with low phosphorus plating solutions and KANIBORON (Ni-P-B), the phosphorus content in the film is low, so 1 gram of nickel sulfate will plate about 2.5 μdm2 (experimental value).
Kajiri (causes of Kajiri)
When the edges of an item have a jagged appearance, as if they have been nibbled on by an animal. (In Japanese it is called "Kajiri")
This phenomenon occurs when some factor causes the composition of the plating solution to be disrupted, resulting in an excess of stabilizer in the bath. This is because stabilizers such as lead contained in the bath do not adhere evenly to the material, but tend to adhere preferentially to specific areas such as the edges.
This can be improved by appropriately adjusting the plating conditions, such as the bath ratio.
In addition to Kajiri, depending on the type of stabilizer used, a faded pattern may also appear in the center rather than the edges.
V/A (bath ratio)
This is a value that indicates the bath load (surface area or number of items put into the plating bath).
The following relationship holds:
V/A = plating tank volume V (cm3) ÷ surface area of items A (cm2)
For general Ni-P plating solutions, it is optimal to use a V/A of 10 to 20.
For example, if you have a 10L (10,000cm3) plating tank, the surface area of items that can be treated is 500 to 1000cm2. For a 10cm square test piece, both sides will be 200cm2, so the number that can be put in is about 2.5 to 5 pieces.
Stabilizers
It is one of the essential components of electroless plating solutions, and is added to suppress the self-decomposition of the bath (a phenomenon in which the reduction reaction of metal ions in the bath progresses in places other than the surface of the product, causing metal components to precipitate).
Generally, if the concentration of the stabilizer is high, the plating bath will be stable, but the plating film will be more likely to occur Kajiri, and if the concentration is low, the plating bath will self-decompose during the reaction, which is one of the causes of roughness, so it is important to manage the concentration in the bath. Our product, Sumer solution, is designed to have the optimal concentration for each type of solution.
Traditionally, lead has been commonly used, but in response to recent tightening environmental laws and regulations, inorganic substances and various organic compounds are also being used as lead substitutes. (SE-660, SFB-27, etc.)
